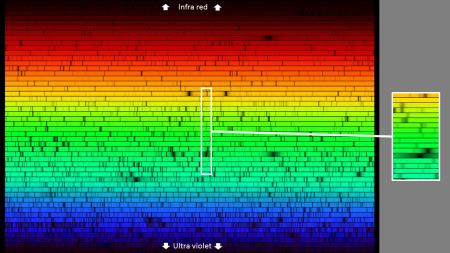
Light through a gas
Visible light travels through most gases almost unperturbed.
And broadly speaking, the Earth’s atmosphere is transparent to visible light.
However if one looks in detail at the way sunlight travels through the Earth’s atmosphere, one can see some remarkable features.
The figure above is a high-resolution spectrum of sunlight. The spectrum would be about 40 times as wide as the figure above but has been ‘folded back’ on itself many times
Light
You may be familiar with the fact that light is a wave in the electric field.
- When the wave vibrates with a frequency of approximately 430 terahertz it has a wavelength of approximately 0.7 thousandths of millimetre, and it elicits the sensation of red in our eyes.
- When the wave vibrates with a frequency of approximately 750 terahertz it has a wavelength of approximately 0.4 thousandths of millimetre, and it elicits the sensation of blue in our eyes.
You are probably familiar with the basic features of the spectrum as it sweeps from light which elicits the sensation of ‘red‘ in our brain, to light which elicits the sensation of ‘blue‘.
But this Figure also shows many dark lines in the spectrum. If we looked at the Sun with filters at these specific frequencies – we would see no light at all! The atmosphere would be opaque!
What has happened is that light with a very specific frequency (and hence wavelength) has been absorbed by vibrations of electrons within specific types of atoms.
Some of these atoms were in the outer layers of the Sun, and some are in our atmosphere.
Infrared Sunlight
Electrical waves exist with lower frequencies that elicit no sensation of colour or brightness in our eyes: this light is called ‘infrared’.
If we look at sunlight coming through the atmosphere at infrared frequencies, the spectrum is even more complex than in the visible region of the spectrum.
The graph below shows data acquired by my colleague Tom Gardiner. It shows the brightness of sunlight coming through the atmosphere at frequencies 10 times lower than visible light.
The brightness is plotted versus the wavelength of the light rather than the frequency because for historical reasons, that is a more common way to present the data.The wavelengths vary between 4 thousandths of a millimetre and 5 thousandths of a millimetre (4 to 5 micrometres).

There are two remarkable things about this spectrum:
- the complexity of the spectrum – there are hundreds of peaks and troughs –
- and the occurrence of a range of wavelengths between about 4.18 and 4.45 micrometres in which the sunlight is completely blocked!
The next two figures show the green region and the orange region in detail.


If one looks at even lower frequencies (longer wavelengths), one sees the same two features – millions of sharp lines and entire ‘blocked bands’ – repeated again and again.
For example, the image below is a modified extract from this amazing image (which I don’t have permission to reproduce) and shows details of transmission of sunlight through the atmosphere at frequencies of around 20 terahertz and wavelengths around 15 micrometres.
This particular range of blocked infrared light is caused by carbon dioxide molecules in the atmosphere. At this range of frequencies the carbon dioxide molecule can bend easily.
Amazingly, this simple ‘bendability’ of the molecule plays a significant role in determining the surface temperature of the Earth.

That’s all for this article:
The story so far is that when one looks up through the atmosphere, there are certain frequencies at which light is blocked.
This blocking sometimes occurs at specific frequencies, and sometimes as ranges of blocked transmission – known as ‘blocked bands’.
For historical and technical reasons, people usually specify the wavelength of the blocked light rather than its frequency.
The next article is about the link between specific blocked frequencies and blocked bands.
January 3, 2017 at 12:22 am |
Great essay!!! Just the thing for the kids in science class who want to know ‘why’ and ‘how’ the ‘greenhouse effect’ works. I’m going to copy it and hand it around to those interested.
January 3, 2017 at 12:23 am |
[…] Making sense of science « 1. Light transmission through the atmosphere […]
January 3, 2017 at 12:58 am |
[…] Michael, why did you write the last four articles (1,2,3,4) on the transmission of infrared radiation through the atmosphere: that stuff is already well […]